Chemical Dominoes
Chemical Dominoes:
In this project we investigated different ways to produce carbon dioxide to blow up a balloon. They discover that mass is conserved in a chemical reaction and learn how to balance chemical equations. They calculate how much chemical they need to blow up a balloon, using magnesium as part of a circuit, students learn about the metal activity series. We learn about the nature of light, phosphorescence, and ground and excited state of electrons. We build a conductivity tester and determine which solutions contain electrolytes. We explore factors that affect reaction rates, and how to control the outcomes. We were challenged to create our own chemical dominoes using the chemical reactions we learned in class. It had to consist of at least 3 reactions.
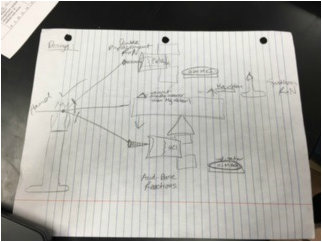
Blue Print:
In this project we investigated different ways to produce carbon dioxide to blow up a balloon. They discover that mass is conserved in a chemical reaction and learn how to balance chemical equations. They calculate how much chemical they need to blow up a balloon, using magnesium as part of a circuit, students learn about the metal activity series. We learn about the nature of light, phosphorescence, and ground and excited state of electrons. We build a conductivity tester and determine which solutions contain electrolytes. We explore factors that affect reaction rates, and how to control the outcomes. We were challenged to create our own chemical dominoes using the chemical reactions we learned in class. It had to consist of at least 3 reactions.
Blue Print:
Chemical Dominoes Video:
|
Chemistry Concepts:
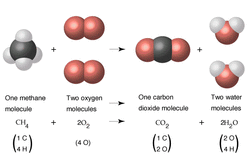
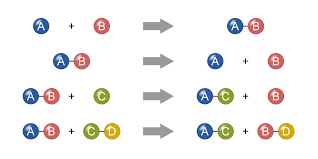
Below is the chemistry behind the four reactions in our chemical dominoes: 1. Combustion Reaction Where: The match burns the convergence of the three ribbons holding the cups with various solutions. Chemical Equation: C2H4(s) + 3O2(g) → 2CO2+ 2H2O(g) Def: a substance combines with oxygen, releasing a large amount of energy in the form of light and heat. This produces carbon dioxide and water vapor 2. Double Displacement Reaction Where: The lever drops lead nitrate into the beaker with potassium iodide. The solution turns yellow. Chemical Formula: 2KI(aq)+ Pb(NO3)2 → 2K(NO3)2 + PbI2(s) About:
3. Synthesis Reaction Chemical Formula: 2Mg(s) + O2(g) → 2MgO(s) Def: When two or more substances combine to form a new compound. → This is NOT a combustion reaction because the two substances form a new complex molecule, not carbon dioxide and water. About:
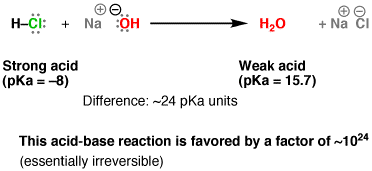
4. Acid-Base Reaction Chemical Formula: NaOH(aq) + HCl(aq) → H2O(l) + NaCl(s) Def: A chemical process in which hydrogen ions are exchanged between either neutral or electrically charged species. About:
|
1.
2.
3.
4.
|
Reflection:
In this project I worked with Zach Muzzarelli, Jessica Walters, and Emilie Baxter. In creating this project we had to think about our Rube Goldberg project from freshman year. We started by creating our blueprint so we could plan out how we would make our chemical dominoes project. We then picked out the reactions we wanted to use in the project. All in all, the planing process was crucial to the creation of the chemical dominoes. I believe my group was pretty creative in how we created it. I loved my group and would love to be with them again in future projects.
While creating this project a lot of planning went into our design. We had to manage our time, and create a blue print and plan out how all the reactions would take place. I believe through this project we improved our time management skills. Through all the components of this project, we didn't fall behind once, and to me this means we improved that skill. Also I believe the knowledge I had about chemistry was improved in this project. I learned about the reactions, and how the components reacted to each other in the way they did. Two disadvantages our group had in this project was based upon our building skills. When creating our levers we failed miserably. For the life of us we couldn't get the levers made. In the end we tried different wood and we created a great final project. Our final disadvantage came in the final test of our chemical dominoes. The lead nitrate we were using wouldn't work, and we found out that it reacts best with the potassium iodine when it was made fresh. So we remade the lead nitrate, and it worked perfectly.
In this project I worked with Zach Muzzarelli, Jessica Walters, and Emilie Baxter. In creating this project we had to think about our Rube Goldberg project from freshman year. We started by creating our blueprint so we could plan out how we would make our chemical dominoes project. We then picked out the reactions we wanted to use in the project. All in all, the planing process was crucial to the creation of the chemical dominoes. I believe my group was pretty creative in how we created it. I loved my group and would love to be with them again in future projects.
While creating this project a lot of planning went into our design. We had to manage our time, and create a blue print and plan out how all the reactions would take place. I believe through this project we improved our time management skills. Through all the components of this project, we didn't fall behind once, and to me this means we improved that skill. Also I believe the knowledge I had about chemistry was improved in this project. I learned about the reactions, and how the components reacted to each other in the way they did. Two disadvantages our group had in this project was based upon our building skills. When creating our levers we failed miserably. For the life of us we couldn't get the levers made. In the end we tried different wood and we created a great final project. Our final disadvantage came in the final test of our chemical dominoes. The lead nitrate we were using wouldn't work, and we found out that it reacts best with the potassium iodine when it was made fresh. So we remade the lead nitrate, and it worked perfectly.